

Olive Shihui Zhu, MS
By Olive Shihui Zhu, MS; Genetic Counselor, ORM Fertility – China
What is Huntington’s Disease
Huntington disease (HD) is an adult-onset neurodegenerative disorder that currently has no cure or preventative treatments available. It is an autosomal dominant condition, meaning that if an individual has HD each of their children has a 50% chance to inherit the HD variant and also develop the disease. Therefore, it is not uncommon to see many generations of a family having affected individuals. HD was one of the first genetic conditions to have its gene sequenced and available for pre-symptomatic predictive genetic testing (screening family members who do not have symptoms of HD but are at risk of developing it based on their family history).
HD is a “trinucleotide repeat” disorder – it is caused by a repetitive sequence of three letters (in this case C-A-G) in a person’s DNA. Testing for HD is done by analyzing the number of CAG repeats in the HD gene, HTT. A count of 6-26 CAG repeats indicates that an individual is not at risk of developing HD. A count of over 40 CAG repeats indicates that an individual will develop HD at some point in their lives; usually, symptoms begin in one’s 30s to 40s and progress for 15 to 20 years. (There is also a gray area of results – between 26-40 CAG repeats – which we will not discuss in this post).
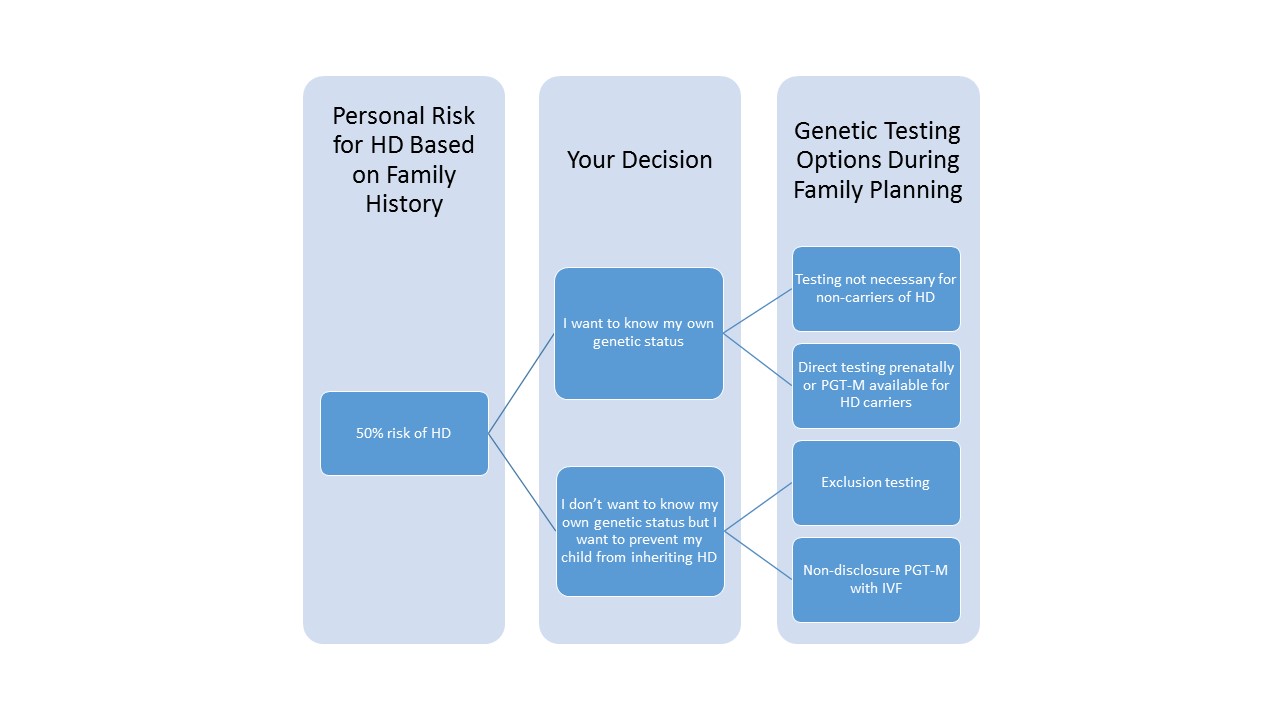
While studying about HD in pursuing my genetic counseling degree I read a novel, Inside the O’Briens, that left a lasting impression on me. The novel exemplified how HD is a family affair and how it affects each family member differently. It illustrated how individuals affected with HD experience slow changes to their health over time due to the progressive symptoms. For this reason, many people who are at 50% risk for HD will find it difficult to decide whether or not to learn their own genetic status through DNA testing. If you are in this situation yourself, I encourage you to speak to a genetic counselor who can guide you through the options. Due to the psychosocial impact of predictive genetic testing for HD for individuals and their family members, international guidelines for predictive testing in HD recommend that there be at least two pre-test counseling sessions prior to taking the test, an in-person appointment for the test, a results and post-test counseling session, and post-test follow up.
If you know that you have inherited the HD allele or if do not know your status but are at 50% risk to have inherited HD, you may be concerned about the risk or possible risk for your offspring to inherit HD. You may not be aware that there is an option that can help you avoid passing the HD gene on to your offspring, even if you do not wish to learn your own HD status.
HD – Huntington disease
IVF – In vitro fertilization
PGT-M – Preimplantation genetic testing for monogenic (single-gene) disorders, previously known as preimplantation genetic diagnosis (PGD)
If you know that you carry the expanded HD gene
If you know that you carry the expanded HD gene, your offspring have a 50% chance to inherit the HD variant. Some people who carry the HD variant will not choose to take any specific measures to avoid having a child who inherits the variant. Those who wish to avoid passing the gene to their offspring, however, may consider getting pregnant on their own and then doing testing of the fetus during pregnancy through prenatal diagnostic procedures such as chorionic villus sampling or amniocentesis. These tests can reveal whether a pregnancy is affected or unaffected. However, these tests are typically pursued only by individuals who would strongly consider terminating an affected pregnancy.
Another alternative – and the only way to avoid conceiving a pregnancy affected with HD – is to pursue preimplantation genetic testing for “monogenic” disease (“PGT-M” – referring to those genetic conditions caused by a change in a single gene). This process is formerly known as preimplantation genetic diagnosis or PGD. In PGT-M, in vitro fertilization (IVF) is performed to create several embryos using the eggs and sperm of the prospective parents. Embryos are grown in an IVF laboratory and after several days of development, a small handful of cells is carefully removed from each embryo for genetic testing. Embryos are frozen while DNA testing is performed. The genetic test determines whether or not embryos have inherited the HD variant, and unaffected embryo(s) are transferred back into the woman’s uterus in the hopes of conceiving a pregnancy. Any resulting child would have a very high chance of being unaffected with HD.
If you are at risk for HD and don’t want to know your status
If you are at risk for HD (you have an affected parent) and do not wish to know your own genetic status but wish to avoid passing HD on to your offspring in case you are a carrier, there other methods of IVF/PGT-M that can be used to test embryos without necessitating that you first learn whether or not you have inherited the HD variant.
The second method is the one more commonly used today: exclusion testing for HD. With this testing, instead of looking at the embryo’s HD gene directly, the DNA fingerprint (in this case, a section of DNA surrounding the HD gene) of the embryo is compared to the DNA of the at-risk parent and their own parents. The test determines whether each embryo inherited the “high-risk” HD allele (this is assumed to be the case if the embryo has the DNA fingerprint from the affected grandparent) or the “low-risk” HD allele (the one from the unaffected grandparent). Only “low-risk” embryos are then transferred into the uterus to result in pregnancy. In order to create this personalized test for each unique family, DNA from both the intended parents and at least one of the grandparents is usually needed. The advantage of this method is that there is no risk of the at-risk parent learning their HD status during the process – neither they nor the embryos are directly tested for the expanded HD allele. The disadvantage is that embryos deemed to be at “high-risk” for HD still have a 50% chance to not carry the expanded HD allele and could actually be unaffected. Therefore, this method can result in having a lower number of embryos that are deemed “safe” for transfer.
Individuals considering PGT-M for HD have many factors to consider. Reproductive genetic counselors are uniquely trained to assist you in understanding your options for preventing the transmission of HD and the benefits and limitations of each choice. If you would like more information about PGT-M for HD or another genetic condition, our team of genetic counselors at ORM would be glad to answer your questions or schedule a consult with you.
References:
de Die-Smulders, C. E. M., De Wert, G. M. W. R., Liebaers, I., Tibben, A., & Evers-Kiebooms, G. (2013). Reproductive options for prospective parents in families with Huntington’s disease: clinical, psychological and ethical reflections. Human reproduction update, 19(3), 304-315.
Losekoot, M., Van Belzen, M. J., Seneca, S., Bauer, P., Stenhouse, S. A., & Barton, D. E. (2013). EMQN/CMGS best practice guidelines for the molecular genetic testing of Huntington disease. European Journal of Human Genetics, 21(5), 480.
MacLeod, R., Tibben, A., Frontali, M., Evers‐Kiebooms, G., Jones, A., Martinez‐Descales, A., … & Editorial Committee and Working Group ‘Genetic Testing Counselling’of the European Huntington Disease Network. (2013). Recommendations for the predictive genetic test in Huntington’s disease. Clinical genetics, 83(3), 221-231.
