Preimplantation Genetic Testing for monogenic/single gene conditions (PGT-M)
Monogenic conditions are those that are caused by mutations in a single gene, rather than by several different genetic and environmental factors. These conditions may have autosomal dominant, autosomal recessive, X-linked, or mitochondrial inheritance. PGT-M is available for the vast majority of serious genetic conditions, as long as the particular gene mutation(s) in the family have been identified through DNA testing. However, the ability to offer PGT-M is dependent not only on the disorder/gene itself but also on the ability to successfully offer a reliable test using available technologies.
What are the steps of the PGT process?
1. Consultation with our team.
2. Genetic testing to identify the specific disease mutation in the family (if not already done)
3. Fertility testing.
4. PGT-M custom test development (often referred to as “making a probe”).
5. In Vitro Fertilization: embryos are created and grown to the blastocyst stage (day 5 or 6).

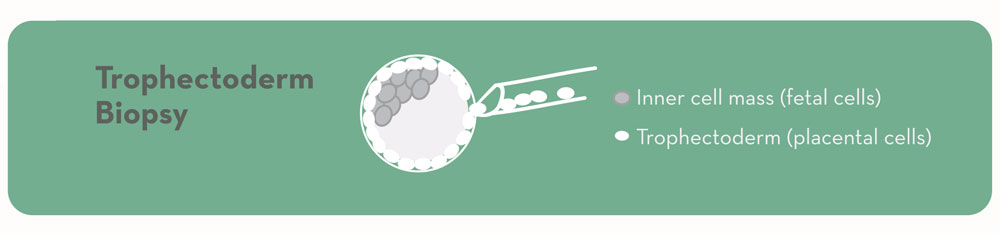
6. Trophectoderm biopsy of ~5-10 cells from the future placental cells of each embryo.
7. Embryos are frozen.
8. Cells removed from the embryos are tested for the single gene condition, as well as for sporadic chromosome problems.

9. Results available.
10. Transfer embryos to uterus of patient or surrogate in a subsequent frozen embryo transfer cycle.
11. Extra embryos remain frozen for future use.
Is the embryo biopsy safe?
In embryos that have developed to day 5 or 6 (blastocysts), we can see the difference between the inner cell mass (cells that will make the baby) and the trophectoderm (cells that will make the placenta). We are able to safely remove cells from the future placenta without disrupting the baby-making cells. The experience and expertise of the embryologists is very important in minimizing risks to the embryos. Less than 1% of the over 10,000 embryos we have biopsied since 2010 did not survive the biopsy. We also believe the long-term risks of blastocyst biopsy to the embryo, pregnancy, and baby to be acceptably low.
Why is the day on which the biopsy is done important?
When genetic testing of embryos first became available, embryos were tested on day 3 at the “cleavage” or 8-cell stage, and only one cell was removed for analysis. Now, biopsy techniques and genetic testing methods have evolved to allow for testing on day 5 or 6 of embryo development. ORM only offers testing at this later stage because it is associated with better implantation rates; the ability to biopsy placental (not fetal) cells; and better reliability of genetic results.
How is genetic testing done in PGT-M?
Genetic testing of embryos is more complicated than genetic testing of, for example, a blood sample. Because only a very small amount of DNA is available, special genetic testing techniques are necessary. Most PGT-M is now done via a technique called karyomapping. Compared to older methods, this technology has improved the accuracy of PGT-M and has also shortened the timeline required to prepare tests for patients.
In karyomapping, DNA samples from the sperm and egg providers, as well as from one or more family member(s) of known genetic status (gene carrier or non-carrier) are examined to identify a unique DNA “fingerprint” that is associated with the gene mutation(s) in the family. (If family members are not available, other special test protocols may be used). Embryos can then be tested to determine whether they carry the DNA fingerprint associated with the “normal” gene or the gene with the mutation.
How do you “make a probe?”
Prior to the IVF cycle, the PGT laboratory undertakes several steps to be sure that an accurate test will be available for embryos. The ability to offer PGT-M is influenced by many factors including the particular disease gene, the family’s specific gene mutation and DNA fingerprint, and the testing technologies that are available. “Probe” is a somewhat outdated term referencing an older technology that required development of an actual, physical DNA probe that was then used to test embryos. Today, the validation of a family’s PGT test prior to IVF is more accurately referred to as the PGT test development process. During this process, the family’s DNA samples are examined to find the DNA fingerprint associated with the disease gene and to determine whether other methods of genetic testing may be necessary in addition to or instead of karyomapping. This ensures – before an IVF cycle is begun – that an accurate test will be possible. The good news is that in the vast majority of cases, a PGT test can be successfully created and the planned IVF cycle can move forward.
What is the accuracy of PGT-M?
Generally the accuracy of PGT-M is >97%, but the specific accuracy may vary depending upon the particular mutation, gene, and/or family being tested. More detailed information about your particular test becomes available from the PGT laboratory following the test development process.
Do you recommend PGT-A (chromosome screening of the embryos) along with PGT-M?
Yes. Due to the high rate of sporadic chromosome abnormalities in embryos (30-80%, depending on maternal age), even from healthy, young, or fertile individuals, PGT-A can provide valuable information and help us find the embryos that are most likely to result in a successful pregnancy. Therefore, ORM always performs PGT-A in conjunction with PGT-M cycles. These tests can be done simultaneously on the same biopsy, and adding PGT-A to a PGT-M IVF cycle does not change the overall process or the timeline for results.
Does PGT replace prenatal testing?
Not necessarily. While transferring an embryo that has tested “normal” by PGT is expected to significantly reduce the chance for the future baby/child to develop the genetic disorder in question, it does not entirely eliminate the risk. Invasive, diagnostic prenatal tests such as chorionic villus sampling (CVS) and amniocentesis are generally considered to be more accurate than PGT (often >99%) and are recommended for those who wish to confirm PGT results in the ongoing pregnancy. Whether or not to have prenatal testing is a personal decision that we recommend discussing in-depth with your prenatal provider and a prenatal genetic counselor.
How do I get started with PGT-M?
We recommend making an appointment for a new patient consultation with one of our physicians to start. You are also more than welcome to email one of our genetic counselors with any initial questions about your specific situation prior to making an appointment.
